- Blog
- I close my eyes
- Imei changer online
- Rising kingdoms multiplayer descargar
- Filmora 9 slow motion
- Program running on this computer is trying to display a message lotus notes client
- Nba 2k15 pc free download
- Berserk anime soundtrack
- Google sketchup pro cost
- Kendrick lamar pimp a butterfly album download
- Nito installer apple tv 3
- Def jam fight for newyork psp
- Nfs most wanted pc cheats pink slip
- What mission is after phil cassidy gta vice city mobile
- Super mario 3d world cemu toad levels
- Vmix playlist
- Pokemon diamond rom zip
- Xj survey remover permission key
- 3ds emulator apk 2016
In view of the toxicity of various Sb forms, biological treatment is inefficient and has not been extensively studied, with notable examples being green marine macroalgae and sulphate-reducing bacteria used for the decontamination of Sb-polluted water 13, 14, 15, 16. Consequently, in order to be used for drinking, such water needs to be treated.Ĭurrently, (electro)chemical (sulphide precipitation, pH adjustment), physicochemical (coagulation, ion exchange, and adsorption), and biological methods are used for the primary treatment of Sb mine wastewater 10, 11, 12. a threshold of 0.005 mg L −1 is set by the GB5749-2006 national standard). well above the maximal values stipulated by drinking water standards (e.g. In the Yangtze River, Sb(III) and Sb(V) concentrations were determined as 0.029–0.736 and 0.121–2.567 mg L −1, respectively 7, i.e. The concentration of dissolved Sb in surface water near mining areas in China ranges from 4.58 to 29.4 μg L −1 8, 9. In nature, Sb occurs in rocks, water, and soil at levels of 0.15–2 mg kg −1, < 1 mg mL −1, and 0.3–8.6 mg kg −1, respectively 4, whereas in the Qingfeng River near the world’s largest Sb mine in the Xikuangshan area, the recorded values was as high as 6384 mg L −1 5, 6, 7. Consequently, Sb pollution is a very serious problem in China. China hosts more than half of the global Sb reserves and is accounted for 100% of the global Sb output from 1998 to 2008 (1.5 × 10 5 tonnes) 3. Consequently, the United States Environmental Protection Agency and the European Union have prioritised the control of this pollutant 2. As a typical heavy metal, Sb is toxic and carcinogenic to humans and other organisms, causing liver, skin, respiratory, and cardiovascular diseases 1. When the pollution level exceeds the limits prescribed by the World Health Organisation and the United States Pharmacopoeia, the pollutants enter the food chain through bioaccumulation and cause serious harm to humans. Water pollution is caused by the direct or indirect entry of untreated contaminants. rGO/NZV-FeZn was suggested to remove Sb(V) through adsorption-photocatalytic reduction and flocculation sedimentation mechanisms and, in view of its high cost performance, stability, and upscalable synthesis, was concluded to hold great promise for source water and wastewater treatment. Column regenerability was tested at an initial concentration of 0.8111 mg Sb L −1, and the treated water obtained after five consecutive runs complied with the GB5749-2006 requirement for Sb. In a practical decontamination test, Sb removal efficiency of 99.38% was obtained for a reaction column filled with 3.5 g of rGO/NZV-FeZn.
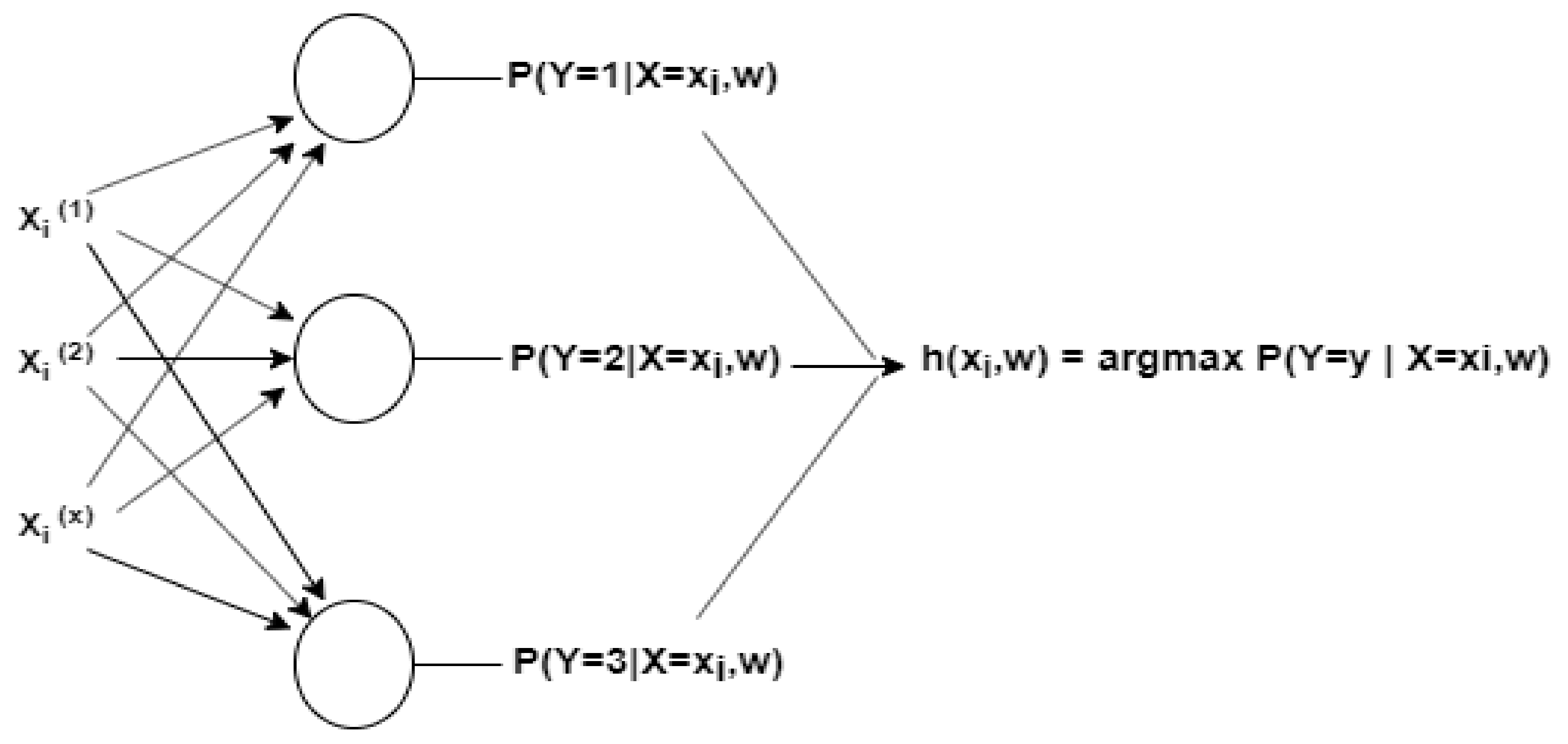
Adsorption data were fitted by a quasi-second-order kinetic equation and Langmuir model, revealing that among these materials, NZV-FeZn exhibited the best Sb removal performance (543.9 mg Sb g NZV-FeZn −1, R 2 = 0.951). In particular, rGO/NZV-FeZn was shown to comprise franklinite, Fe 0, and graphite. Herein, weakly magnetic nano-Fe–Zn materials (NZV-FeZn, GAC SP/NZV-FeZn, and rGO/NZV-FeZn) capable of rapid and efficient Sb(V) adsorption from water were prepared and characterised. In order to remove antimony(V) from water, the purpose of this study was to prepare a novel graphene nano iron zinc (rGO/NZV-FeZn) photocatalyst via hydrothermal method followed by ultrasonication.

The existence of toxic and carcinogenic pentavalent antimony in water is a great safety problem.
- Blog
- I close my eyes
- Imei changer online
- Rising kingdoms multiplayer descargar
- Filmora 9 slow motion
- Program running on this computer is trying to display a message lotus notes client
- Nba 2k15 pc free download
- Berserk anime soundtrack
- Google sketchup pro cost
- Kendrick lamar pimp a butterfly album download
- Nito installer apple tv 3
- Def jam fight for newyork psp
- Nfs most wanted pc cheats pink slip
- What mission is after phil cassidy gta vice city mobile
- Super mario 3d world cemu toad levels
- Vmix playlist
- Pokemon diamond rom zip
- Xj survey remover permission key
- 3ds emulator apk 2016
